|
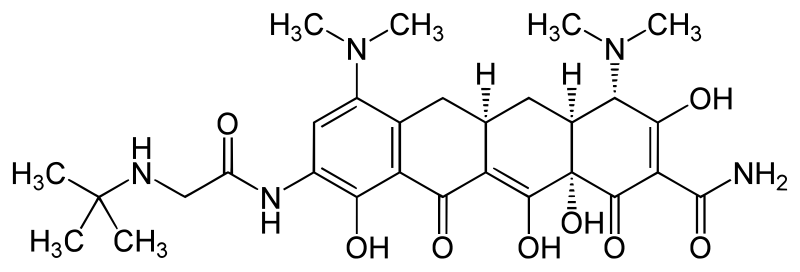
N-[(5aR,6aS,7S,9Z,10aS)-9-[amino(hydroxy)methylidene]-4,7-bis(dimethylamino)-1,10a,12-trihydroxy-8,10,11-trioxo-5,5a,6,6a,7,8,9,10,10a,11-decahydrotetracen-2-yl]-2-(tert-butylamino)acetamide
|
|
Distribution
Plasma protein binding ranges from 71% to 89%. Vd is 7 to 9 L/kg.
Metabolism
Not extensively metabolized.
Elimination
The t ½ is approximately 42 h (multiple dosing). Primary route of elimination is biliary/fecal excretion (59% of dose); glucuronidation and renal excretion are secondary routes (33% of dose excreted in urine, 22% as unchanged tigecycline).
Special Populations
Hepatic Function Impairment
Systemic Cl reduced 25% and t ½ prolonged 23% in patients with moderate hepatic function impairment (Child-Pugh class B); systemic Cl reduced 55% and t ½ prolonged 43% in patients with severe hepatic function impairment (Child-Pugh class C).
|
|
Contraceptives, oral
Coadministration may decrease the effectiveness of the oral contraceptive.
Warfarin
Because warfarin Cl may be reduced and plasma levels may be increased, monitor PT or other suitable anticoagulation parameters.
|







































