| 結構式 |
|
|
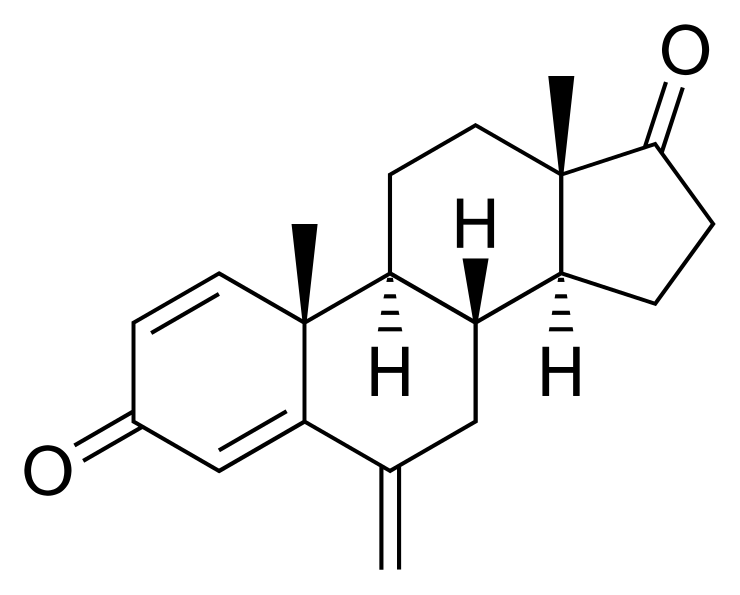
10,13-dimethyl-6-methylidene- 7,8,9,10,11,12,13,14,15,16- decahydrocyclopenta[a] phenanthrene- 3,17-dione
|
| UpToDate |
UpToDate 連結
|
| 藥理作用 |
|
Exemestane就是藉由抑制芳香環轉化酶而阻斷雄性素轉化成雌激素降低了體內雌激素的濃度,達到抑制乳癌細胞擴散的目的。值得注意的是,其抑制的機轉稱為自殺性抑制方式,由exemestane和芳香環轉化酶不可逆的結合使芳香環轉化酶失去活性。
這個機轉可以解釋為何exemestane停藥一段時間,體內雌激素的濃度仍然很低的原因。
|
| 適應症 |
|
|
具有雌激素受體陽性之停經婦女,使用Tamoxifen至少2年之早期侵犯性乳癌的輔助治療。已接受抗雌激素而無效之停經婦女晚期乳癌。
|
| 用法用量 |
|
|
口服,推薦劑量為一次25 mg,1日1次,宜飯後服用。採用AROMASIN的治療應堅持直至腫瘤進展。對肝腎功能不全患者無需劑量調整。
|
| 藥動力學 |
|
|
Absorption
Rapidly absorbed. At least 42% if the dose is absorbed. T max is 1.2 h (women with breast cancer) and 2.9 h (healthy women). When taken after a high-fat breakfast, the plasma levels increased approximately 40%.
Distribution
90% protein bound.
Metabolism
Extensively metabolized. CYP3A4 is the principal isoenzyme involved in the oxidation of exemestane. The metabolites are inactive or inhibit aromatase with decreased potency compared to exemestane.
Elimination
The t 1/2 is 24 h. Approximately 42% is excreted in the urine and approximately 42% is excreted in the feces. Less than 1% is excreted unchanged in the urine.
Special Populations
Renal Function Impairment
AUC is about 3 times higher in those with moderate or severe renal insufficiency. No dosage adjustment necessary.
Hepatic Function Impairment
AUC increased approximately 3 times in those with moderate or sever hepatic insufficiency. No dosage adjustment necessary.
|
| 副作用 |
|
|
最常見的是面部潮紅和噁心。其他常見的不良事件是疲勞、出汗增加和頭暈。報導較少見的不良事件中,發生率>=2%的有頭痛、失眠、疼痛、皮疹、腹痛、厭食、嘔吐、抑鬱、脫髮、全身或下肢水腫、便秘和消化不良。
|
| 交互作用 |
|
Agents that inhibit or induce CYP-4503A4 (eg, rifampin, phenobarbital, erythromycin, ketoconazole, and others)
A possible decrease of exemestane by agents which inhibit or induce CYP-450 3A4 (eg, rifampin, phenobarbital, erythromycin, ketoconazole).
|
| 禁忌 |
|
|
對藥物或任何輔料成份過敏者、停經前婦女、懷孕或哺乳婦女。
|
| 給付規定 |
|
9.1.1.Exemestane (如Aromasin Sugar Coated Tablets):(88/11/1、90/10/1、99/6/1、105/8/1)
1.限停經後或卵巢切除後,且女性荷爾蒙受體 (estrogen receptor) 陽性之晚期乳癌病患,經使用tamoxifen 無效後,方可使用。
2.具有雌激素受體陽性之停經婦女,使用tamoxifen 至少兩年之高危險早期侵犯性乳癌的輔助治療,且不得與tamoxifen 或其他aromatase inhibitor 併用。使用時需同時符合下列規定:(105/8/1)
(1)病歷上應詳細記載手術資料、病理報告(應包含ER、PR 之檢測結果且無復發現象)。
(2)本案藥品使用不得超過三年。
|
| 注意事項 |
|
1.處於停經前內分泌狀態的婦女不可使用,臨床使用前應通過評估LH、FSH和Estradiol來確認婦女處於停經後狀態。
2.運動員慎用。
|
| 藥品保存方式 |
|
|
藥品應置於攝氏 15 ~ 25 度乾燥處所;如發生變質或過期,不可再食用。
|







































